Preimplantation genetic testing (PGT) is an established reproductive option for couples with a risk of conceiving a baby that is affected with some known genetic disease. The aim of PGT is to have an unaffected child and in turn avoiding the trauma of terminating the pregnancy. This method allows embryo testing for embryos being produced via in vitro fertilization (IVF) before transferring the embryo into the uterus.

Types of PGT
There are two types of genetic testing of an embryo prior to transfer:
Preimplantation Genetic Screening (PGT-A) and Preimplantation Genetic Diagnosis (PGT-M).
PGT-A
Preimplantation genetic testing for aneuploidy (PGT-A) serves as a screening procedure to identify embryo with chromosomal abnormality, which is thought to be the most common reason IVF treatment fails. For example, Down syndrome is the most common chromosome disorder that we know of which carries an additional chromosome 21.
Genetic testing for abnormal chromosome number is known as Preimplantation Genetic Testing for Aneuploidies (PGT-A).
- Older women who are trying to conceive
- Recurrent miscarriages
- Achieving live birth at the shortest time
So, is PGT-A recommended for every couple regardless of women’s age?
The answer is a big resounding NO. The data collected by Society of Assisted Reproductive Technology (2015) revealed that PGT-A is only helpful for women aged 35 years and above which further proves that women who are older have lower chance of pregnancy and/or delivering a healthy baby.
PGT-M
In contrast, Preimplantation genetic testing for monogenic/single gene disorder (PGT-M) refers specifically to when one or both genetic parents have a known genetic abnormality and testing is performed on an embryo to determine if it also carries the genetic abnormality.
Because only unaffected embryos are transferred to the uterus for implantation, PGT-M provides an alternative to current post-conception diagnostic procedures (i.e. amniocentesis or chorionic villus sampling), which are frequently followed by the difficult decision of pregnancy termination if results are unfavourable. PGT-M is presently the only option available to avoid having a child affected with a genetic disease prior to implantation. It is an attractive means of preventing heritable genetic disease, thereby eliminating the dilemma of pregnancy termination following unfavourable prenatal diagnosis.
Genetic testing to diagnose embryos with known genetic disorder is now known as Preimplantation Genetic Testing for Monogenic Disorder (PGT-M).
if a man and/or woman has a known genetic disorder (eg : Thalassemia) and are trying to conceive, they can opt for PGT-M to reduce the risk of pregnancy with the inherited genetic disorder.
How is PGT performed?
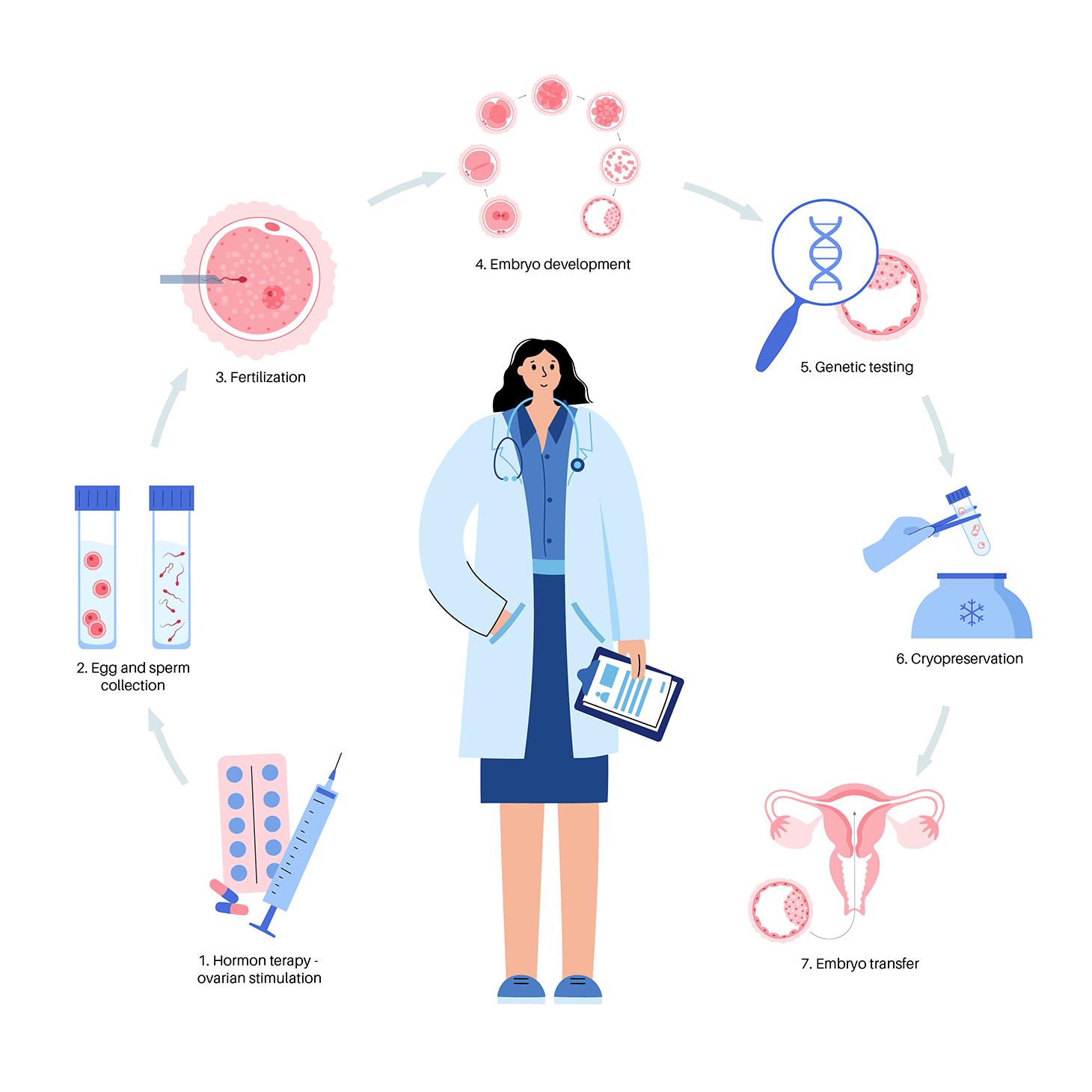
Risks of PGT
- Risk of embryo biopsy – embryos may be damaged during the biopsy procedure or may fail to survive
- No embryos for biopsy – not all embryos will develop into blastocysts are suitable for biopsy
- Risk of embryo freezing – not all the embryos will survive freeze/thaw process
- Possibility of misdiagnosis due to mosaicism
- No results or inconclusive results
- Possibility of no embryos for transfer due to unfavourable results
- Unpredictable and uncontrolled problems with transportation of biopsied cells to external molecular laboratory, such as weather or accident that are beyond control
If you are interested in knowing more or have decided to get in touch with us, please call us at
Or send us your enquiry